
Global Parkinson’s Disease Drugs Market Forecasts to 2024
- July, 2018
- Domain: Healthcare - Pharmaceuticals
- Get Free 10% Customization in this Report
Overview: Parkinson’s disease is one of the most prevalent neurodegenerative disorders, which progresses with age because of damaged dopaminergic cells in the brain. Bradykinesia, rigidity, postural instability, and tremors are the symptoms of the Parkinson’s disease. Huge patient pool and rising rate of diagnosis are the significant factors driving the market growth. The Parkinson’s Disease Foundation has stated that nearly 10 million people are suffering from this disorder globally. The highest frequency of this disorder is observed in developed countries, and in the US, almost 60,000 new cases are reported annually.
PD is an idiopathic disease for which the cause of the occurrence is still unknown, hence only symptomatic drugs are available in the market for the treatment. Furthermore, the disorder is diagnosed in the final stages when the majority of the cells that produce dopamine lose their functionality. This is demanding more innovative and novel drugs for the treatment.
Market Analysis: The “Global Parkinson’s Disease Drugs Market” is estimated to witness a CAGR of 7.2% during the forecast period 2018–2024. The market is analyzed based on two segments – drug class and regions.
Regional Analysis: The regions covered in the report are North America, Europe, Asia Pacific, and Rest of the World (RoW). North America accounts for the largest share of the market, followed by Europe, Asia Pacific, and Rest of the World. More than 40% of the market is occupied by Europe, with Germany being the major contributor to the market growth.
Drug Class Analysis: Based on drug classes, the market is segmented into levodopa, MAO-B inhibitors, COMT inhibitors, dopamine agonists, anticholinergics, and others. Levodopa is the most attractive drug in the market owing to its highly effective treatment in controlling the motor symptoms throughout the stages of Parkinson’s disease. Recently, in January 2018, Adamas Pharmaceuticals Inc. launched Gocovri for the treatment of dyskinesia in individuals with PD, who are taking levodopa. This is the original drug indicated explicitly for dyskinesia, which is technologically advanced with the long-term use of levodopa.
Key Players: The major players in the market include Novartis, Roche, AbbVie, Teva Pharmaceutical, Pfizer, STADA Arzneimittel AG, GSK, Valeant, Merck & Co. Inc., and Amneal Pharmaceuticals Inc.
Competitive Analysis: New product launch and collaboration are the primary strategies being followed by the market players to maintain their leadership and strengthen the market position. In January 2015, Neuropore Therapies Inc. and UCB S.A. signed a commercialization agreement for NPT200-11. Recently, Impax Pharmaceuticals’ RYTARY and AbbVie’s DUOPA received the US FDA approval for the treatment of Parkinson’s disease. Other strategies include business partnerships, mergers & acquisitions, and a multifaceted pipeline. Recently, in January 2018, Adamas Pharmaceuticals Inc. launched Gocovri for the treatment of dyskinesia in individuals with PD, who are taking levodopa.
Benefits: The report provides complete details about the usage and adoption rate of Parkinson’s disease drugs in various regions. With that, key stakeholders can know about the major trends, drivers, investments, and vertical player’s initiatives. Moreover, the report provides details about the major challenges that are going to impact on the market growth. Additionally, the report gives the complete details about the key business opportunities to key stakeholders to expand their business and capture the revenue in the specific verticals to analyze before investing or expanding the business in this market.
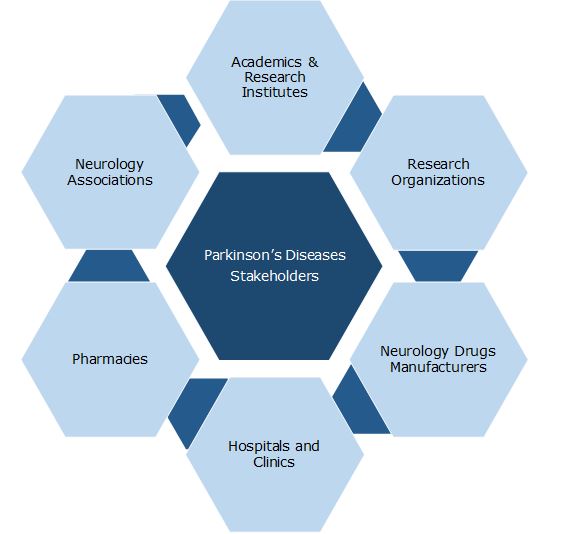
Key Stakeholders:
1 Industry Outlook
1.1 Industry Overview
1.1.1 Global Driver for Pharmaceutical Demand:
1.1.2 Pharmaceutical Spending Region Wise
1.1.3 R&D Pipeline in Pharmaceutical Industry
1.1.4 Top Pharma Drugs by Sales in 2017 ($Million)
1.2 Total Addressable Market
2 Report Outline
2.1 Report Scope
2.2 Report Summary
2.3 Research Methodology
2.4 Report Assumptions
3 Market Snapshot
3.1 Market Definition – Infoholic Research
3.2 Segmented Addressable Market (SAM)
3.3 Trends of Parkinson Diseases Market
3.4 Related Markets
3.4.1 Neurosurgical Devices
3.4.2 Neurointerventional Devices
3.4.3 Over the Counter Drugs (OTC)
4 Market Outline
4.1 Pipeline Products of Parkinson Diseases Drugs
4.2 Market Segmentation
4.3 Porter 5 (Five) Forces
4.4 PEST Analysis
5 Market Characteristics
5.1 Market Dynamics of Parkinson Disease Drugs Market
5.1.1 Drivers
5.1.1.1 Increasing aging population
5.1.1.2 Increasing prevalence of Parkinson disease
5.1.2 Opportunities
5.1.2.1 Supportive initiatives from government and other organizations
5.1.2.2 Exhaustive pipeline and increasing clinical trials
5.1.3 Restraints
5.1.3.1 Expiry of patents for blockbuster drugs and availability of generic drugs
5.1.3.2 Complex drug development process
5.1.4 DRO – Impact Analysis
5.1.5 Key Stakeholders
6 Drug Type: Market Size and Analysis
6.1 Overview
6.2 Levodopa
6.3 Dopamine agonists
6.4 MAO-B inhibitors
6.5 COMT Inhibitors
6.6 Anticholinergics
6.7 Others
7 Regions: Market Size and Analysis
7.1 Overview
7.2 North America
7.2.1 Overview
7.3 Europe
7.3.1 Overview
7.4 APAC
7.4.1 Overview
7.5 Rest of the World
7.5.1 Overview
8 Competitive Landscape
9 Vendor Profiles
9.1 Novartis AG
9.1.1 Overview
9.1.2 Business Units
9.1.3 Geographic Presence
9.1.4 Business Focus
9.1.5 SWOT Analysis
9.1.6 Business Strategy
9.2 GlaxoSmithKline Plc
9.2.1 Overview
9.2.2 Geographic Presence
9.2.3 Business Focus
9.2.4 SWOT Analysis
9.2.5 Business Strategy
9.3 F. Hoffmann-La Roche Ltd.
9.3.1 Overview
9.3.2 Business Units
9.3.3 Geographic Presence
9.3.4 Business Focus
9.3.5 SWOT Analysis
9.3.6 Business Strategy
9.4 AbbVie Inc.
9.4.1 Overview
9.4.2 Geographic Presence
9.4.3 Business Focus
9.4.4 SWOT Analysis
9.4.5 Business Strategies
9.5 Merck & Co., Inc.
9.5.1 Overview
9.5.2 Business Units
9.5.1 Geographic Presence
9.5.2 Business Focus
9.5.3 SWOT Analysis
9.5.4 Business Strategies
9.6 Teva Pharmaceutical Industries Ltd.
9.6.1 Overview
9.6.2 Business Units
9.6.3 Geographic Presence
9.6.4 Business Focus
9.6.5 SWOT Analysis
9.6.6 Business Strategy
9.7 Valeant Pharmaceuticals International Inc.
9.7.1 Overview
9.7.2 Business Units
9.7.3 Geographic Presence
9.7.4 Business Focus
9.7.5 SWOT Analysis
9.7.6 Business Strategy
10 Companies to Watch For
10.1 Boehringer Ingelheim GmbH
10.1.1 Overview
10.1.2 Highlights
10.2 Amneal Pharmaceuticals Inc.
10.2.1 Overview
10.3 Wockhardt
10.3.1 Overview
10.3.2 Highlights
10.4 STADA Arzneimittel AG
10.4.1 Overview
10.4.2 Highlights
10.5 Pfizer Inc.
10.5.1 Overview
10.5.2 Highlights
10.6 Sun Pharmaceuticals Industries Ltd.
10.6.1 Overview
10.6.2 Highlights
Annexure
Abbreviations
Research Framework
Infoholic research works on a holistic 360° approach in order to deliver high quality, validated and reliable information in our market reports. The Market estimation and forecasting involves following steps:
- Data Collation (Primary & Secondary)
- In-house Estimation (Based on proprietary data bases and Models)
- Market Triangulation
- Forecasting

Market related information is congregated from both primary and secondary sources.
Primary sources
involved participants from all global stakeholders such as Solution providers, service providers, Industry associations, thought leaders etc. across levels such as CXOs, VPs and managers. Plus, our in-house industry experts having decades of industry experience contribute their consulting and advisory services.
Secondary sources
include public sources such as regulatory frameworks, government IT spending, government demographic indicators, industry association statistics, and company publications along with paid sources such as Factiva, OneSource, Bloomberg among others.
