
Global Non-invasive Prenatal Testing (NIPT) Market Forecast to 2025
- March, 2019
- Domain: Healthcare - Diagnostics
- Get Free 10% Customization in this Report
[83 pages report] This market research report includes a detailed segmentation of the global non-invasive prenatal testing market – By Application (Trisomy, Microdeletion Syndrome, and Other NIPT Applications), and By Region (North America, Europe, Asia Pacific, and Rest of the World).
Overview of the Non-invasive Prenatal Testing Market Research
Infoholic’s market research report predicts that the global non-invasive prenatal testing market will grow at a CAGR of 17.5% during the forecast period 2019–2025. The market has witnessed steady growth in the past few years and development in technology with the introduction of advanced products has increased the acceptance of non-invasive prenatal testing products in the market. The market is fueled by an upsurge in the number of newborns with chromosomal disorders due to the increasing number of late pregnancies and growing demand for non-invasive procedures for early diagnosis.
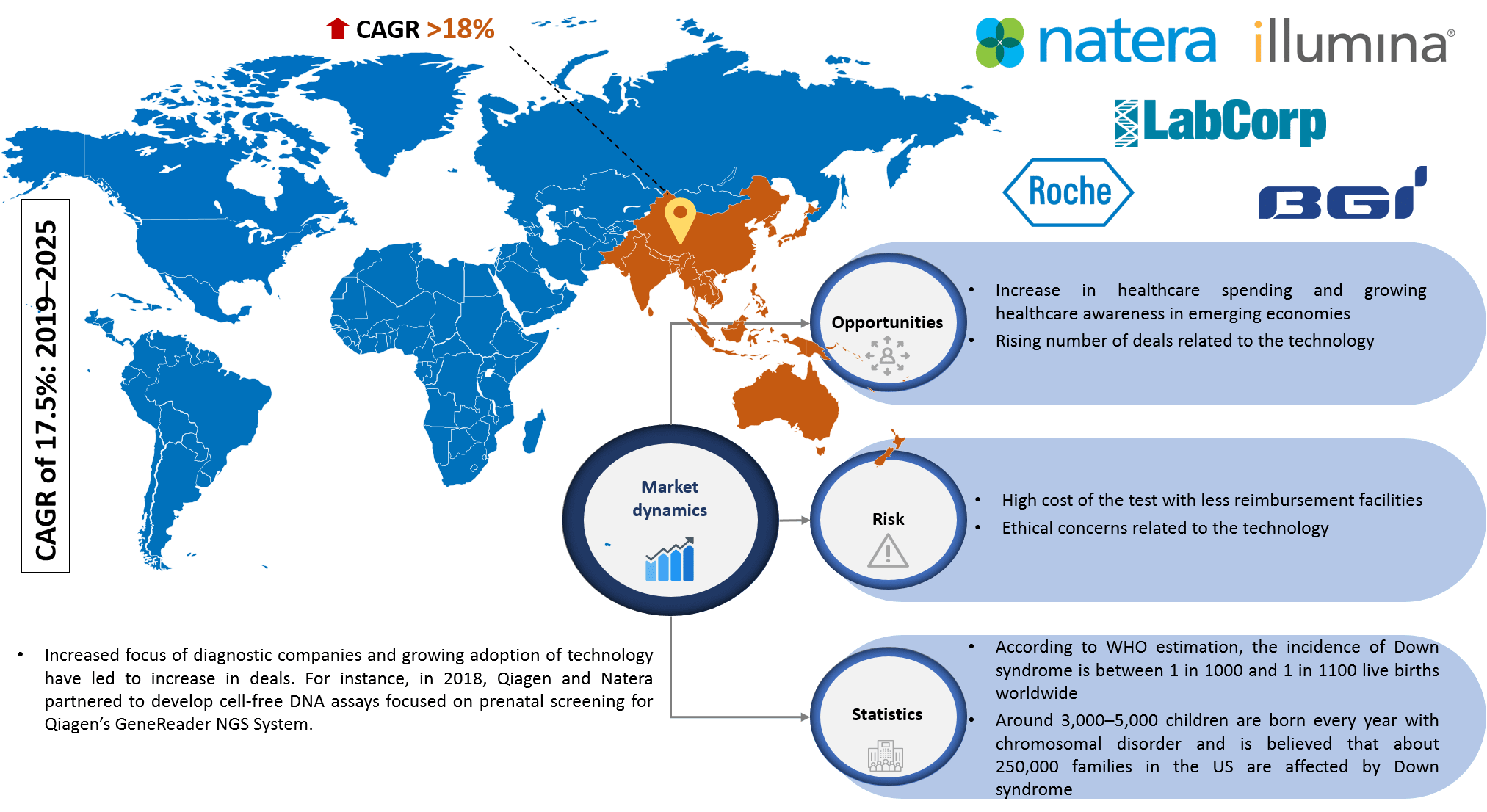
The global market continues to grow, and NIPT is among the most widely used techniques for detecting genetic disorders in the fetal stage, which will help in the proper management of disease during the initial period of the gestation. The field is getting revolutionized with the advancement in technology. Vendors are focusing on new product launches, product approvals, and targeting end-user's perspective. The market generates revenue from the key players operating in this field, and few of them include Illumina Inc., Laboratory Corporation of America Holdings, Natera Inc., F. Hoffmann-La Roche Ltd., and Beijing Genomics Institute.
According to Infoholic Research analysis, North America accounted for the largest share in the global non-invasive prenatal testing market in 2018 and will retain a high position during the forecast period. According to WHO estimation, the prevalence of Down syndrome is between 1 in 1,000 and 1 in 1,100 live births worldwide. The incidence of Down syndrome is 1 in 212 with the women age between 35 and 39 and the risk significantly increases with maternal age, i.e., above 45, the incidence rate is 1 in 33 newborns. But the risk rate is very low in younger women in the age group of 24–25 years with incidence rate reduced to 1 in 1429. The risk of aneuploidy also increases with maternal age with an incidence rate of 1 in 192 for women in the age group of 35 years and 1 in 66 in the age group up of 40. Favorable reimbursement policies, availability of advanced technologies, presence of prominent vendors, and an increase in the prevalence of genetic disorders will increase the adoption and demand for tests in this region. Asia Pacific is expected to grow at a high CAGR during the forecast period, owing to factors such as increasing awareness of early disease diagnosis, increasing healthcare expenditure, and presence of huge population base.
Non-invasive Prenatal Testing Market by Application:
- Trisomy
- Microdeletion Syndrome
- Other NIPT Applications
In 2018, the trisomy segment occupied the largest share and is expected to grow at a high CAGR during the forecast period. Increasing incidence of chromosomal anomalies coupled with advanced maternal age makes the segment the major shareholder in the global non-invasive prenatal testing market. Further, innovations in technology are likely to propel the growth of the segment during the forecast period.
Non-invasive Prenatal Testing Market by Regions:
- North America
- Europe
- APAC
- RoW
The global market is dominated by North America, followed by Europe and Asia Pacific. The US occupied the dominant share in the North American market due to the increasing adoption of high- and average-risk NIPT test, rising number of late pregnancies that increase the number of newborns with Down syndrome, no risk of miscarriage, and availability of the advanced tests due to the presence of dominant vendors in the region.
Non-Invasive Prenatal Testing Market Research Competitive Analysis – The global non-invasive prenatal testing market has massive growth opportunities in both developed and developing regions. The advancements of tests will increase the competition among vendors. The increased focus of diagnostics and biotechnology companies has led to an increase in deals related to non-invasive testing products due to its increasing popularity. 10x Genomics Inc. and Berry Genomics Co. Ltd. entered into a collaboration to further develop approaches for next-generation NIPT in January 2018. In early 2018, IONA test developed by Premaitha Health plc has been approved by Brazil's regulatory authority, Agência Nacional de Vigilância Sanitária, for sale in the country as an IVD. In late 2018, PerkinElmer’s Vanadis NIPT system obtained CE-IVD mark for commercialization and distribution in Europe and in other countries where CE-IVD mark is accepted. The key market vendors are focusing on increasing the product portfolio. There are few acquisitions in the last few years. Premaitha acquired Yourgene Bioscience in Taiwan in March 2017. In July 2017, Eurofins Scientific closed the acquisition of GATC Biotech AG and this transaction comprises the acquisition of 62.63% of the shares owned by GATC in LifeCodexx AG, one of the Europe’s specialists in NIPT. In addition, other leading players are focusing on hugely investing in R&D activities to develop new products to attain a maximum share in the market.
Key vendors:
- Natera Inc.
- Laboratory Corporation of America Holdings (LabCorp)
- Illumina Inc.
- Hoffmann-La Roche
- Beijing Genomics Institute
- LifeCodexx AG
- Quest Diagnostics
- Yourgene Health Plc
- Myriad Genetics Inc.
- NIPD Genetics
- PerkinElmer Inc.
- Next Biosciences (Pty) Limited
Key competitive facts:
- The market is highly competitive with all the players competing to gain market shares. Intense competition, rapid advancements in technology, frequent changes in government policies, and the prices are key factors that confront the market.
- The requirement of high initial investment, implementation, and maintenance cost in the market are also limiting the entry of new players.
Benefits – The report provides complete details about the adoption rate of non-invasive prenatal tests. Thus, the key stakeholders can know about the major trends, drivers, investments, vertical player’s initiatives, and government initiatives in the upcoming years along with details of the pureplay companies entering the market. Moreover, the report provides details about the major challenges that are going to impact the market growth. Additionally, the report gives complete details about the key business opportunities to key stakeholders in order to expand their business and capture the revenue in specific verticals, and to analyze before investing or expanding the business in this market.
Key Takeaways:
- Understanding the potential market opportunity with precise market size and forecast data.
- A detailed market analysis focusing on the growth of the non-invasive prenatal testing
- Factors influencing the growth of the non-invasive prenatal testing
- In-depth competitive analysis of dominant and pure-play vendors.
- Prediction analysis of the non-invasive prenatal testing industry in both developed and developing regions.
- Key insights related to major segments of the non-invasive prenatal testing
- Latest market trend analysis impacting the buying behavior of the consumers.
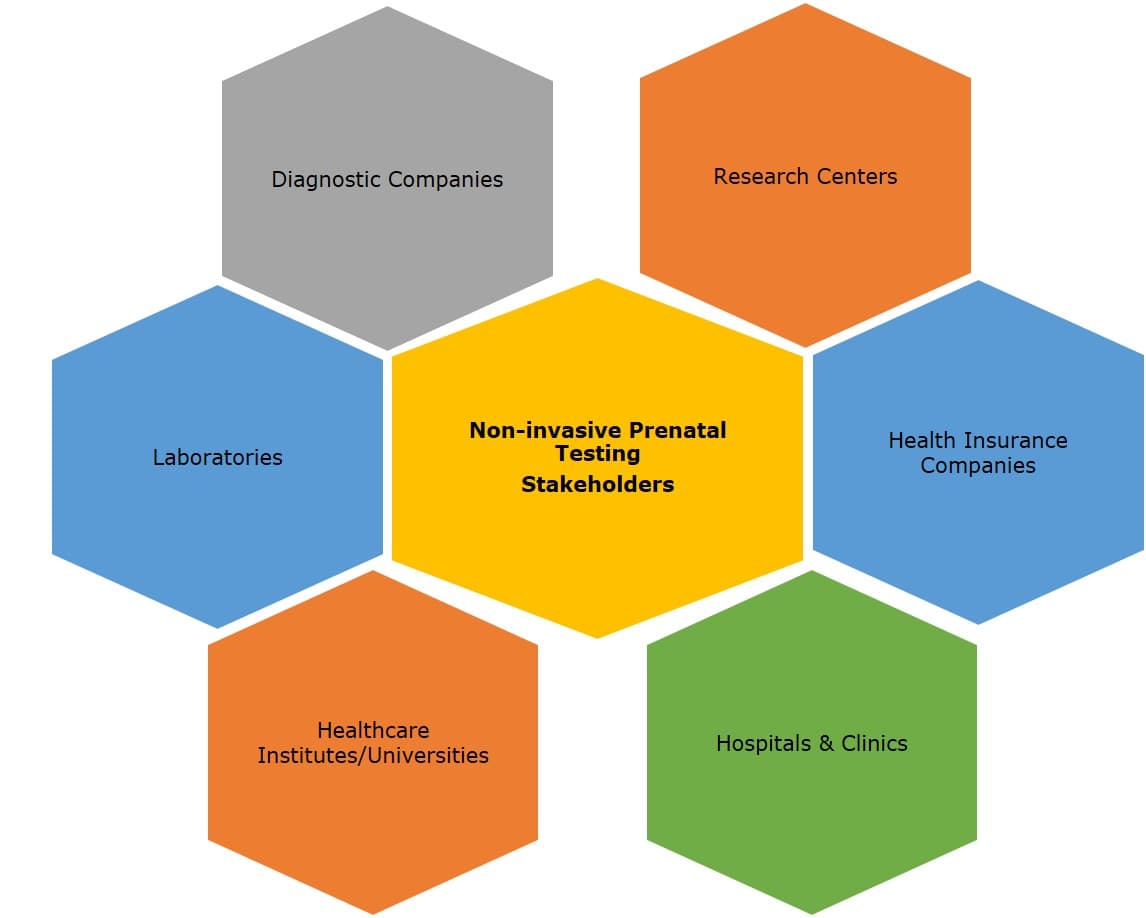
Key Stakeholders
1 Industry Outlook
1.1 Industry Overview
1.2 Total Addressable Market
1.3 Industry Trends
2 Report Outline
2.1 Report Scope
2.2 Report Summary
2.3 Research Methodology
2.4 Report Assumptions
3 Market Snapshot
3.1 Market Definition – Infoholic Research
3.2 Device Benefits
3.3 Segmented Addressable Market
3.4 Trends in the Non-invasive Prenatal Testing Market
3.5 Related Markets
3.5.1 Genetic Testing
3.5.2 Cell-free DNA Testing
3.5.3 Transplantation Diagnostics
3.5.4 Liquid Biopsy
4 Market Outlook
4.1 Market Segmentation
4.2 PEST Analysis
4.3 Porter 5(Five) Forces
5 Market Characteristics
5.1 DRO – Market Dynamics
5.1.1 Drivers
5.1.1.1 Upsurge in the Number of Babies with Chromosomal Disorders due to Increasing Number of Late Pregnancies
5.1.1.2 Growing Demand for Non-Invasive Procedures for Early Diagnosis
5.1.2 Opportunities
5.1.2.1 Increase in Healthcare Spending and Growing Healthcare Awareness in Emerging Economies
5.1.2.2 Rising Number of Deals Related to the Technology
5.1.3 Restraints
5.1.3.1 High Cost of the Test with Less Reimbursement Facilities
5.1.3.2 Ethical Concerns Related to the Technology
5.2 DRO – Impact Analysis
5.3 Key Stakeholders
6 Applications: Market Size and Analysis
6.1 Overview
6.2 Trisomy
6.3 Microdeletion Syndrome
6.4 Other NIPT Applications
7 Regions: Market Size and Analysis
7.1 Overview
7.2 North America
7.3 Europe
7.4 Asia Pacific
7.5 Rest of the World
8 Competitive Landscape
8.1 Overview
9 Vendor Profiles
9.1 Natera Inc.
9.1.1 Overview
9.1.2 Geographic Presence
9.1.3 Business Focus
9.1.4 SWOT Analysis
9.1.5 Business Strategies
9.2 Illumina Inc.
9.2.1 Overview
9.2.2 Business Units
9.2.3 Geographic Presence
9.2.4 Business Focus
9.2.5 SWOT Analysis
9.2.6 Business Strategies
9.3 Laboratory Corporation of America Holdings (LabCorp)
9.3.1 Overview
9.3.2 Business Units
9.3.3 Geographic Revenue
9.3.4 Business Focus
9.3.5 SWOT Analysis
9.3.6 Business Strategies
9.4 F. Hoffmann-La Roche Ltd.
9.4.1 Overview
9.4.2 Business Units
9.4.3 Geographic Revenue
9.4.4 Business Focus
9.4.5 SWOT Analysis
9.4.6 Business Strategies
9.5 Beijing Genomics Institute
9.5.1 Overview
9.5.2 Business Focus
9.5.3 SWOT Analysis
9.5.4 Business Strategies
10 Companies to Watch For
10.1 Quest Diagnostics
10.1.1 Overview
10.2 LifeCodexx AG
10.2.1 Overview
10.3 Yourgene Health Plc
10.3.1 Overview
10.4 NIPD Genetics
10.4.1 Overview
10.5 Myriad Genetics Inc.
10.5.1 Overview
10.6 PerkinElmer Inc.
10.6.1 Overview
10.7 Next Biosciences (Pty) Limited
10.7.1 Overview
11 Annexure
11.1 Abbreviations
TABLE 1 FUNDING SCENARIO 18
TABLE 2 GLOBAL NON-INVASIVE PRENATAL TESTING MARKET REVENUE BY REGIONS, 2018–2025 ($MILLION) 34
TABLE 3 OTHER PROMINENT VENDORS IN THE NON-INVASIVE PRENATAL TESTING MARKET 41
TABLE 4 NATERA INC.: OFFERINGS 42
TABLE 5 NATERA INC.: RECENT DEVELOPMENTS 42
TABLE 6 ILLUMINA INC.: PRODUCT OFFERINGS 47
TABLE 7 ILLUMINA INC.: RECENT DEVELOPMENTS 47
TABLE 8 LABORATORY CORPORATION OF AMERICA HOLDINGS: OFFERINGS 56
TABLE 9 LABORATORY CORPORATION OF AMERICA HOLDINGS: RECENT DEVELOPMENTS 56
TABLE 10 F. HOFFMANN-LA ROCHE LTD.: PRODUCT OFFERINGS 61
TABLE 11 F. HOFFMANN-LA ROCHE LTD.: RECENT DEVELOPMENTS 61
TABLE 12 BEIJING GENOMICS INSTITUTE: PRODUCT OFFERINGS 67
TABLE 13 BEIJING GENOMICS INSTITUTE: RECENT DEVELOPMENTS 67
TABLE 14 QUEST DIAGNOSTICS: SNAPSHOT 70
TABLE 15 QUEST DIAGNOSTICS: RECENT DEVELOPMENTS 70
TABLE 16 LIFECODEXX AG: SNAPSHOT 73
TABLE 17 LIFECODEXX AG: RECENT DEVELOPMENTS 73
TABLE 18 YOURGENE HEALTH PLC: SNAPSHOT 75
TABLE 19 YOURGENE HEALTH PLC: RECENT DEVELOPMENTS 75
TABLE 20 NIPD GENETICS: SNAPSHOT 76
TABLE 21 NIPD GENETICS: RECENT DEVELOPMENTS 76
TABLE 22 MYRIAD GENETICS INC.: SNAPSHOT 77
TABLE 23 MYRIAD GENETICS INC.: RECENT DEVELOPMENTS 77
TABLE 24 PERKINELMER INC.: SNAPSHOT 79
TABLE 25 PERKINELMER INC.: RECENT DEVELOPMENTS 79
TABLE 26 NEXT BIOSCIENCES (PTY) LIMITED: SNAPSHOT 80
TABLE 27 NEXT BIOSCIENCES (PTY) LIMITED: RECENT DEVELOPMENTS 80
Research Framework
Infoholic Research works on a holistic 360° approach in order to deliver high quality, validated and reliable information in our market reports. The Market estimation and forecasting involves following steps:
- Data Collation (Primary & Secondary)
- In-house Estimation (Based on proprietary data bases and Models)
- Market Triangulation
- Forecasting

Market related information is congregated from both primary and secondary sources.
Primary sources
Involved participants from all global stakeholders such as Solution providers, service providers, Industry associations, thought leaders etc. across levels such as CXOs, VPs and managers. Plus, our in-house industry experts having decades of industry experience contribute their consulting and advisory services.
Secondary sources
Include public sources such as regulatory frameworks, government IT spending, government demographic indicators, industry association statistics, and company publications along with paid sources such as Factiva, OneSource, Bloomberg among others.






