
Global Biologics Market Drivers, Opportunities, Trends, and Forecasts 2024
- May, 2018
- Domain: Healthcare - Pharmaceuticals
- Get Free 10% Customization in this Report
Biologics drugs are complex molecules that are manufactured from living organisms or components of living organisms. Biologics include a wide variety of products derived from human, animal, or microorganisms by using biotechnological processes. Many biologics are produced using recombinant DNA technology. They are sometimes referred to as biopharmaceuticals or biological drugs. Various types of biologic drugs include monoclonal antibody, recombinant proteins/hormones, vaccines, cell and gene therapy and others. These biologics may contain proteins that control the action of other proteins and cellular processes, genes that control production of vital proteins, modified human hormones, or cells that produce substances that suppress or activate components of the immune system. Biologics are sometimes referred as referred to as biologic response modifiers, as they change the manner of operation of natural biologic intracellular and cellular actions.
The market for biologics is driven by increasing incidence of disease across the globe, increasing number of biologics approvals and emergence of next generation biologics such as cell and gene therapy. Strong pipelined products and huge number of ongoing clinical trials are providing market growth oppurtunity. Challenges associated with biologics production, high cost of the drug, increasing number of biologics going off patent and emergence of biosimilars are hampering market growth.
Market Analysis: The “Global Biologics market” is estimated to witness a CAGR of 9.9% during the forecast period 2018–2024. The global market is analyzed based on three segments – Product, Application and regions.
Regional Analysis: The regions covered in the report are the North America, Europe, Asia Pacific, and Rest of the World (ROW). North America is the major shareholder in the global biologics market, followed by Europe. North America dominates the biologics market due to increasing product approvals, increasing demand for the product to treat various disease conditions, and presence of established players in this region. Asia-Pacific region is expected to have the fastest growth rate during the forecasted period due to increasing research activities, flexible regulatory environment for clinical trials, growing awareness about biopharmaceutical therapeutics, and rising healthcare expenditure.
Product Analysis: The market by product is segmented into monoclonal antibodies, recombinant proteins/hormones, vaccines, cell and gene therapy and others. Monoclonal antibodies occupied major market share of global biologics market in 2017 and cell and gene therapy is expected to grow at high CAGR during the forecasted period. Wide application of monoclonal antibodies in treatment of various diseases makes this segment, a major shareholder of biologics products market.
Application Analysis: The market by application is segmented into cancer, infectious disease, immunological disorders, hematological disorders, cardiovascular disease and others. Among various application, cancer occupied the largest share in 2017 and is expected to grow at fastest rate during the forecasted period. Growing elderly population, changing lifestyle, increasing disease prevalence makes cancer, the fastest growing application segment during the forecasted period.
Key Players: F. Hoffmann-La Roche, Amgen, Inc., Sanofi S.A., Novo Nordisk A/S, Johnson and Johnson, AbbVie, Inc., Novartis AG, GlaxoSmithKline plc, Bristol-Myers Squibb, Merck & Co., Eli Lilly & Company Ltd., AstraZeneca PLC, Teva Pharmaceutical Industries Ltd., Takeda Pharmaceutical Company and other predominate and niche players.
Competitive Analysis: Currently cancer segment dominates the global biologics segment. Biologics are entering new therapeutic areas such as, asthma and allergy where they are not present historically. The key market players are taking the advantage of strategic deals and product approvals to increase their share in the market. For instance, in September 2017, GSK and AbCellera Biologics, Inc. entered an antibody discovery research collaboration, for the discovery of monoclonal antibodies against an undisclosed membrane protein target. In August 2017, Novartis received first FDA approval from FDA for CAR-T cell therapy, Kymriah to treat patients with B-cell precursor acute lymphoblastic leukemia (ALL) and in May 2018, Novartis received second FDA approval for Kymriah to treat patients with B-cell lymphoma.
Benefits: The report provides complete details about the usage and adoption rate of biologics in various therapeutic verticals and regions. With that, key stakeholders can know about the major trends, drivers, investments, and vertical player’s initiatives. Moreover, the report provides details about the major challenges that are going to impact on the market growth. Additionally, the report gives the complete details about the key business opportunities to key stakeholders to expand their business and capture the revenue in the specific verticals to analyze before investing or expanding the business in this market.
Key Stakeholders: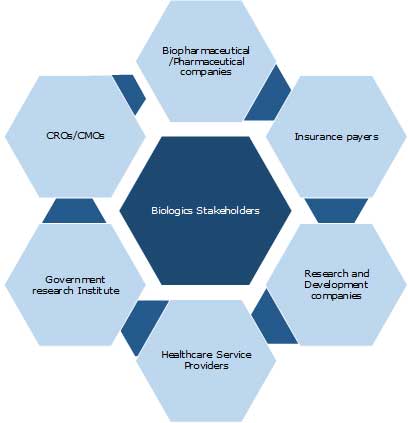
Report Scope:
- Product Type
- Monoclonal antibodies
- Recombinant Proteins
- Vaccines
- Cell therapy
- Gene therapy
- Others
- Source
- Microbial source
- Mammalian source
- Application
- Cancer
- Infectious disease
- Haematological disorder
- Immunological disorder
- Others
- Regions
- North America
- Europe
- Asia Pacific
- Rest of the World
1 Industry Outlook
1.1 Industry Overview
1.2 Industry Trends
1.2.1 R&D Pipeline in the Pharmaceutical Industry
2 Report Outline
2.1 Report Scope
2.2 Report Summary
2.3 Research Methodology
2.4 Report Assumptions
3 Market Snapshot
3.1 Market Definition – Infoholic Research
3.2 Segmented Addressable Market
3.3 Trends in the Biologics Market
3.4 Related Markets
3.4.1 Over-the-counter Drugs
3.4.2 Orphan Drugs
3.4.3 Human Insulin
4 Market Outlook
4.1 Market segmentation
4.2 PEST Analysis
4.3 Porter 5(Five) Forces
5 Market Characteristics
5.1 DRO Analysis – Market Dynamics
5.1.1 Drivers
5.1.1.1 Growing incidence of disease across the globe
5.1.1.2 Increasing number of biologics approvals
5.1.2 Opportunities
5.1.2.1 Strong portfolio of product pipeline and huge number of ongoing clinical trials
5.1.2.2 Increasing opportunities in emerging markets
5.1.3 Restraints
5.1.3.1 Challenges associated with biologics production
5.1.3.2 Increasing number of biologics going off patent and emergence of biosimilars
5.2 DRO – Impact Analysis
5.3 Key Stakeholders
6 Products: Market Size and Analysis
6.1 Overview
6.2 Monoclonal Antibodies
6.3 Recombinant Proteins/Hormones
6.4 Vaccines
6.5 Cell & Gene Therapy
6.6 Others
7 Applications: Market Size and Analysis
7.1 Overview
7.2 Cancer
7.3 Immunological Disorders
7.4 Infectious Disease
7.5 Hematologic Disorders
7.6 Cardiovascular Disease
7.7 Others
8 Regions: Market Size and Analysis
8.1 Overview
8.2 North America
8.2.1 US
8.2.2 Canada
8.3 Europe
8.3.1 UK
8.3.2 Germany
8.3.3 France
8.3.4 Spain
8.4 Asia Pacific
8.4.1 India
8.4.2 China
8.4.3 Japan
8.5 Rest of the World
9 Competitive Landscape
10 Vendors Profile
10.1 F. Hoffmann-La Roche
10.1.1 Overview
10.1.2 Business Units
10.1.3 Geographic Presence
10.1.4 Business Focus
10.1.5 SWOT Analysis
10.1.6 Business Strategies
10.2 Sanofi S.A.
10.2.1 Overview
10.2.2 Business Units
10.2.3 Geographic Presence
10.2.4 Business Focus
10.2.5 SWOT Analysis
10.2.6 Business Strategies
10.3 Amgen Inc.
10.3.1 Overview
10.3.2 Geographic Presence
10.3.3 Business Focus
10.3.4 SWOT Analysis
10.3.5 Business Strategies
10.4 Novo Nordisk A/S
10.4.1 Overview
10.4.2 Business Units
10.4.3 Geographic Presence
10.4.4 Business Focus
10.4.5 SWOT Analysis
10.4.6 Business Strategies
10.5 AbbVie Inc.
10.5.1 Overview
10.5.2 Geographic Presence
10.5.3 Business Focus
10.5.4 SWOT Analysis
10.5.5 Business Strategies
10.6 Johnson & Johnson
10.6.1 Overview
10.6.2 Business Units
10.6.3 Geographic Revenue
10.6.4 Business Focus
10.6.5 SWOT Analysis
10.6.6 Business Strategies
10.7 Novartis AG
10.7.1 Overview
10.7.2 Business Units
10.7.3 Geographic Revenue
10.7.4 Business Focus
10.7.5 SWOT Analysis
10.7.6 Business Strategies
11 Companies to Watch for
11.1 Bristol–Myers Squibb
11.1.1 Overview
11.2 GlaxoSmithKline plc
11.2.1 Overview
11.3 Merck & Co.
11.3.1 Overview
11.4 Eli Lilly and Company Ltd.
11.4.1 Overview
11.5 Teva Pharmaceutical Industries Ltd.
11.5.1 Overview
11.6 AstraZeneca PLC
11.6.1 Overview
11.7 Takeda Pharmaceuticals
11.7.1 Overview
Annexure
Abbreviations
TABLE 1 PHARMA DRUGS BY SALES, 2017 13
TABLE 2 GLOBAL BIOLOGICS MARKET REVENUE BY REGIONS, 2017–2024 ($MILLION) 49
TABLE 3 OTHER PROMINENT VENDORS OF BIOLOGICS MARKET 59
TABLE 4 F. HOFFMANN-LA ROCHE: OFFERINGS 60
TABLE 5 F. HOFFMANN-LA ROCHE: RECENT DEVELOPMENTS 60
TABLE 6 SANOFI.: OFFERINGS 66
TABLE 7 SANOFI: RECENT DEVELOPMENTS 67
TABLE 8 AMGEN INC.: OFFERINGS 73
TABLE 9 AMGEN INC.: RECENT DEVELOPMENTS 74
TABLE 10 NOVO NORDISK A/S: OFFERINGS 81
TABLE 11 NOVO NORDISK A/S: RECENT DEVELOPMENTS 82
TABLE 12 ABBVIE INC.: OFFERINGS 88
TABLE 13 ABBVIE INC: RECENT DEVELOPMENTS 88
TABLE 14 JOHNSON & JOHNSON: PRODUCT OFFERINGS 92
TABLE 15 JOHNSON & JOHNSON: RECENT DEVELOPMENTS 93
TABLE 16 NOVARTIS AG: PRODUCT OFFERINGS 99
TABLE 17 NOVARTIS AG: RECENT DEVELOPMENTS 100
TABLE 18 NOVARTIS AG: OVERVIEW SNAPSHOT 102
TABLE 19 BRISTOL-MYERS SQUIBB: SNAPSHOT 106
TABLE 20 BRISTOL-MYERS SQUIBB: RECENT DEVELOPMENTS 107
TABLE 21 GLAXOSMITHKLINE PLC.: OVERVIEW 108
TABLE 22 GLAXOSMITHKLINE PLC: RECENT DEVELOPMENTS 109
TABLE 23 MERCK & CO: OVERVIEW 110
TABLE 24 MERCK & CO: RECENT DEVELOPMENTS 110
TABLE 25 ELI LILLY AND COMPANY: SNAPSHOT 112
TABLE 26 ELI LILLY AND COMPANY: RECENT DEVELOPMENTS 112
TABLE 27 TEVA PHARMACEUTICAL INDUSTRIES LTD.: OVERVIEW 113
TABLE 28 TEVA PHARMACEUTICAL: RECENT DEVELOPMENTS 113
TABLE 29 ASTRAZENECA PLC: SNAPSHOT 115
TABLE 30 ASTRAZENECA PLC: RECENT DEVELOPMENTS 115
TABLE 31 TAKEDA PHARMACEUTICALS: SNAPSHOT 117
TABLE 32 TAKEDA PHARMACEUTICALS: RECENT DEVELOPMENTS 118
Research Framework
Infoholic Research works on a holistic 360° approach in order to deliver high quality, validated and reliable information in our market reports. The Market estimation and forecasting involves following steps:
- Data Collation (Primary & Secondary)
- In-house Estimation (Based on proprietary data bases and Models)
- Market Triangulation
- Forecasting

Market related information is congregated from both primary and secondary sources.
Primary sources
Involved participants from all global stakeholders such as Solution providers, service providers, Industry associations, thought leaders etc. across levels such as CXOs, VPs and managers. Plus, our in-house industry experts having decades of industry experience contribute their consulting and advisory services.
Secondary sources
Include public sources such as regulatory frameworks, government IT spending, government demographic indicators, industry association statistics, and company publications along with paid sources such as Factiva, OneSource, Bloomberg among others.






